Anavex (AVXL): Sometimes you just never know where to start with a company
Just an epic dumpster fire
There are certain raging dumpster fires where you just don’t know where to begin. Anavex is one of them. I’m probably not going to break much new ground here but with a $500 million market cap, I just can’t resist to write something about it.
Anavex is developing their lead program blarcamesine (Anavex 2-73) for a whole host of indications including Alzheimer’s Disease, Parkinson’s Disease and Rett Syndrome.
With regards to Rett Syndrome, the company announced on June 6th that they had completed dosing of patients in their 92-patient study. That was over 4 months ago and we still have not seen data. To give you a sense, usually it doesn’t take more than a month or two after the completion of a trial for a CRO to confirm all the data with the sites and get it to the company.
This is not the first time there was weirdness around an Anavex trial nor even the first time with weirdness around a trial conducted by Anavex in Rett Syndrome. Back in February 2022, the company announced data from a 33-patient Phase 3 that they claim was positive but only because they changed the endpoints two weeks before releasing the data! While changing endpoints can be legitimate, usually you don’t see it happen 4 months after the completion of the study and two weeks before releasing data.
One thing you’ll notice is that they changed the Rett Syndrome Behavior Questionnaire (RSBQ) endpoint from the usual change from baseline endpoint to one that uses the non-standard “drug exposure-dependent response”. They did the same thing with the Clinical Global Impressions - Global Improvement (CGI-I) endpoint. Note that Acadia got an FDA approval in Rett thanks to change from baseline data in RSBQ and CGI-I.
Worst part is that even after these endpoint changes, the p-value was still a borderline p=0.037 in both endpoints. Which brings me to another point, you usually don’t see the exact same data in two different endpoints. In fact, I don’t think I’ve ever seen it. Needless to say, it’s pretty clear they failed that Rett trial.
These shenanigans don’t stop with the Rett program, they pulled something similar with their Alzheimer’s data where they changed endpoints a couple weeks after seeing the last patient on their last visit.
Once again, they changed it from a relatively simple change from baseline endpoint to a version of a responder analysis. So you get sentences like “patients treated with ANAVEX®2-73 were 84% more likely to have improved cognition by ADAS-Cog score change of -0.50 points or better from baseline to end of treatment than patients on placebo” in the announcement. It’s just never a good sign when you have to re-read a sentence from a data release to figure out what the heck is being said.
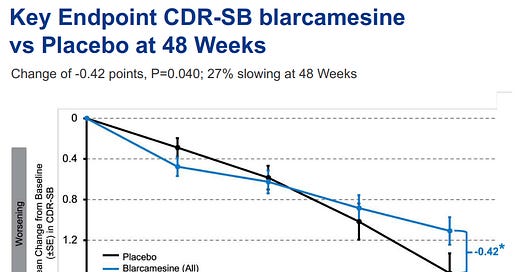
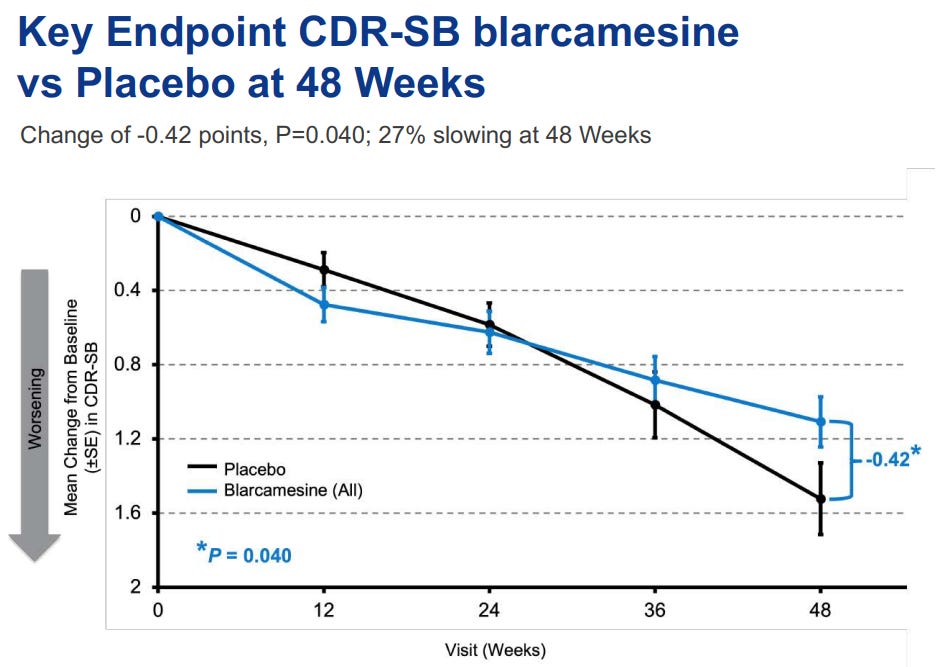
Of course, they did actually disclose the results of change from baseline in CDR-SB, which is an approvable endpoint, but the chart really isn’t reassuring:
This is not what an efficacious drug looks like. Usually treated patients don’t do worse for 6 months and then suddenly start doing better at some random timepoint in the fugure. Here is what the Leqembi chart looks like for the same endpoint, as you can see immediate separation with no bouncing back and forth:
Additionally, they had tested two different doses of the drug, 30mg and 50mg, and yet only presented pooled data, which means to me that even with the endpoint changes, neither dose itself would be statistically significant.
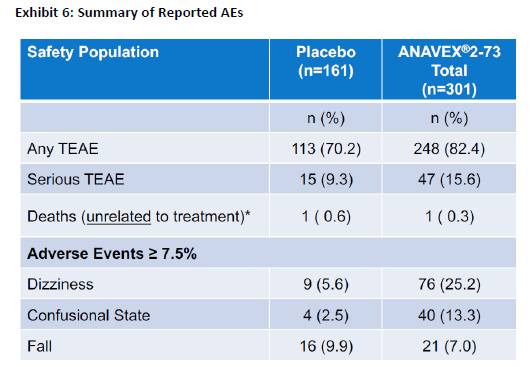
If after all that you think this is an approvable drug and not an abject failure, here is another chart for you, the safety:
A quarter of the patients have dizziness and 13% have a confusional state? Do you really want to give such a drug to someone with Alzheimer’s? Or Parkinson’s? I think that the FDA would have some major issues with this.
I could go on and on with this company, it’s really had a long history of shenanigans. Like here is a chart from a slide presentation from 2009:
Even when they were a preclinical company, they were trying to pump the stock with these outlandish claims. Where’s the $6 billion in sales they promised?
Another issue which was pointed out here, is that the company, for some reason, changed headquarters seven times in an eight year period. These included locals from British Columbia to Greece. Why would they need to do that?
I could probably write a book on these guys so I’ll stop here. We’ll see what entertainment they provide when (if) they release the Rett data.
Please like, share and subscribe!
Stocks mentioned: